This came up a little while ago in my comments section on another post. It was mentioned that things treated with ethylene oxide should be avoided by people with glycol allergies. Specifically the claim was made that ethylene oxide was used to sterilize spices, and thus spices should be avoided. I thought I’d investigate this one a bit further.
What is ethylene oxide?
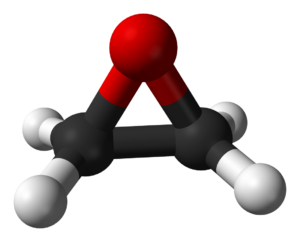
Ethylene Oxide
Ethylene oxide is a colourless, reactive, flammable gas. It is reacted with water and a catalyst to make ethylene glycols, and chemically converted to make products like fibres, foils, antifreeze, plasticizer, solvents, cosmetics, bottles, and sports equipment. Ethylene oxide is both very toxic and a carcinogen, as well as being anaesthetic and a mutagen. Its molecule is essentially a triangle with four legs – an oxygen atom sits on top, with two carbon atoms connected to both the oxygen atom and each other. Each carbon atom is also connected to two hydrogen atoms. As a gas, it leaves no residue on the items it touches, which is why it is also used as a sterilizer for medical equipment.
Should I be concerned about ethylene oxide cross-reacting with ethylene glycols?
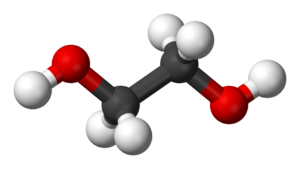
Ethylene Glycol
Allergic cross-reaction requires similarly-shaped molecules; similar enough that the body can’t tell the difference. In the case of ethylene glycol and ethylene oxide, the key word for molecular similarity isn’t the first word – it’s the second. Ethylene only means that there are two carbon atoms with two hydrogen atoms attached to each. It’s the “oxide” and “glycol” distinction that allow us to differentiate the very different structures. Whereas ethylene oxide was a triangle with legs, ethylene glycols are a variable-length, zig-zaggy chain, with an oxygen atom connected to a hydrogen atom at each end. There is no way that the body could confuse those two molecular structures.
What about cross-contamination?
It’s true that any chemical reacted to make a product will still exist in the finished product in its original form in some quantity. In this case, ethylene oxide is used to make ethylene glycols, and thus a small quantity of ethylene oxide will exist in ethylene glycols. The same is not true of the reverse. Ethylene glycols will not exist in unreacted ethylene oxide. There is no risk of cross-contamination in that direction.
So is ethylene oxide safe then?
 Ethylene oxide may not be an allergic danger to glycol allergy sufferers, but it is still an extremely toxic chemical. Where it exists, even in small quantities, there is the possibility of harm to an individual. This includes ethylene glycols and plastics made from glycols. Any unreacted ethylene oxide on the surface of the material should evaporate quickly after production and be of no concern. Fortunately, in places where ethylene oxide gas has been used to disinfect, there is little danger of any harm from the ethylene oxide, as the gas does not deposit itself on surfaces at temperatures above -111 degrees Celsius (-168 degrees Fahrenheit) or warmer.
Ethylene oxide may not be an allergic danger to glycol allergy sufferers, but it is still an extremely toxic chemical. Where it exists, even in small quantities, there is the possibility of harm to an individual. This includes ethylene glycols and plastics made from glycols. Any unreacted ethylene oxide on the surface of the material should evaporate quickly after production and be of no concern. Fortunately, in places where ethylene oxide gas has been used to disinfect, there is little danger of any harm from the ethylene oxide, as the gas does not deposit itself on surfaces at temperatures above -111 degrees Celsius (-168 degrees Fahrenheit) or warmer.
Ultimately it is up to each person to decide what chemicals they choose to include in their lives. Luckily, since it evaporates and becomes a gas far below any temperatures seen outside of a laboratory or industrial environment, this one should not make a noticeable appearance in the daily lives of most people.

Have you come across any information about how PGI is used by utility companies, gas company specifically?
I had forgotten about that one. I believe I did respond to it (can’t remember at the moment), but yes, I can address it in more detail in the future – I’ll make a note. I have direct experience in the natural gas industry and family ties, so I know which practices are fairly uniform across the industry and which are company-specific, and well as understanding how odourant is used and typical gas composition. It’s going to be a busy month for me, but I’ll try to get that done. Stay tuned.
Will do thanks?
Hi, I have tested for high exposure to ethylene oxide. A few questions….
I owned a boutique and dealt with clothing and plastic wraps all day. I also had new furniture deliveries monthly. Curious if it could have come from this?
We moved into a new home when this started. Could be coincidence but curious where it could come from in a home? I did purchase slot of new for this home.
Now that I’ve been exposed and toxic. How do I detox? Infrared Sauna?
A few more questions: How is it you were tested for exposure to ethylene oxide? Did someone say it was stored in your body? Did you have some sort of single toxic exposure that resulted in a hospital visit? If so, what were you doing at the time? Can you give a few more details?